THE National Agency for Food and Drug Administration and Control (NAFDAC) issued a public health warning on Thursday evening regarding the circulation of a “counterfeit” malaria medication.
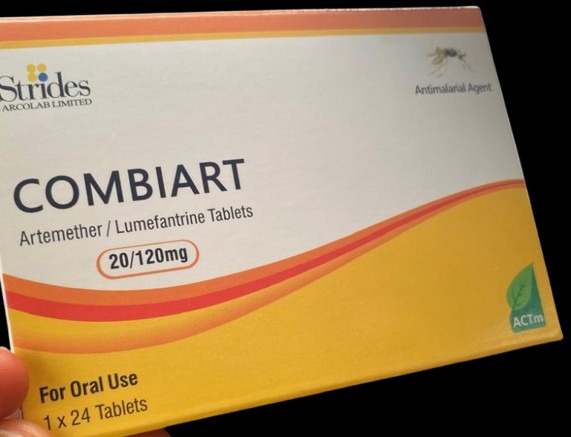
The drug, Combiart Dispersible Tablets (20/120 mg), “is manufactured by Strides Arcolab Limited, based in India. The product was discovered in FCT and Rivers State during surveillance activities,” NAFDAC said in a series of posts on X.
Strides Arcolab Limited has not reacted to the development.
The drug contains zero active ingredients, rendering it ineffective in treating malaria, NAFDAC added.
Malaria is a life-threatening disease caused by parasites that are transmitted to people through the bites of infected mosquitoes, according to WHO.
The drug agency said the registration number on the drug packet was “incorrect” and that the drugs were manufactured between February and June 2023.
“Healthcare professionals and consumers are advised to report any suspicion of the sale of substandard and falsified medicines or medical devices to the nearest NAFDAC office,” NAFDAC urged on X.
The agency stressed the drug lacks proper quality control, potentially leading to serious health consequences.
The drug also fails to effectively treat illnesses like malaria, allowing the disease to worsen, NAFDAC warned.
Trending
- Iran agrees to reopen Strait of Hormuz, halt operations under two-week ceasefire deal
- Dangote refinery raises petrol to N1,275, diesel to N1,950 amid global oil pressure
- ADC chairmen reject Mark, Bala factions, back INEC’s derecognition
- Resident doctors suspend infinite strike, gives FG 14 days ultimatum
- Trump warns Iran could be destroyed ‘by tomorrow night’ if no deal is reached
- Kemi Badenoch tops UK poll as most popular party leader
- Tinubu approves ₦3.3 Trillion payment to resolve power sector debts
- Burkina Faso must forget democracy; it is not for us, says Ibrahim Traoré
- Fani-Kayode criticizes Peter Obi’s Abacha comments, declares he will never be President”
- Wike under fire over PDP unity claim ahead of 2027